
Cancer treatment is no longer defined based on the cancer location only, but increasingly through the genetic and molecular signature, forcing researchers to reshape traditional clinical trials to be more precise and patient profile specific, thus evolving precision medicine.
Master protocols are designs used to improve precision medicine trials through assessing multiple interventions for one cancer type or assessing single or multiple interventions in different diseases.
Basket trial and umbrella trial designs are the most common master protocols used; despite being different, both play a critical role in evolving precision medicine, reducing time for answering multiple questions by combining all in one study.
What is a basket trial?
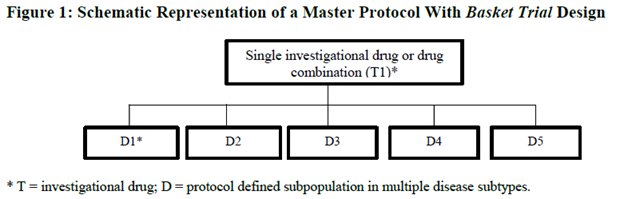
Basket trial is an innovative clinical trial design that is gaining popularity in oncology. It replaces the traditional ways of conducting a clinical trial that compares a therapeutic drug in one cancer type to standard therapy. Instead, patients are grouped according to shared molecular alterations, and a single targeted therapy is evaluated across multiple tumour types.
Basket trials enable healthcare professionals to select the treatment based on the genetic and molecular profile of patients instead of using a particular regimen for a specific cancer type. For example, it allows using vemurafenib in the treatment of cancers that include the BRAF V600 mutation, not only melanoma.
What is the umbrella trial?
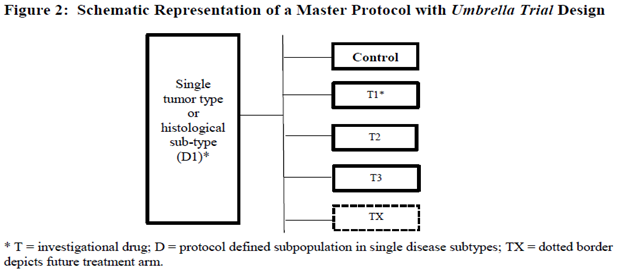
A single cancer type can be a result of a variety of genetic mutations, or biomarkers, for example, Non-Small Cell Lung Cancer (NSCLC), in which molecular heterogeneity is significant, can be caused by EGFR Mutation, KRAS Mutation (G12C), PD-L1 Expression, or other mutations. In this case, an umbrella trial protocol is used to evaluate multiple interventions in the same cancer type in parallel.
The umbrella trial design is when several subtrials are conducted simultaneously, and patients are assigned to different arms based on their mutation/biomarker. Still, all share the same type of cancer.
Strengths and limitations
Basket trials allow researchers to evaluate a single targeted intervention across multiple cancer types that share the same molecular alteration, making them particularly valuable in rare cancers and uncommon genetic mutations. By combining multiple treatment evaluations within one protocol, they may reduce redundancy and improve cost-efficiency compared with traditional trial designs. However, because basket trials focus on specific biomarker-defined subgroups, their findings may have limited generalizability to the broader cancer population. In addition, small sample sizes and the complexity of trial execution may affect statistical power and operational feasibility.
Umbrella trials enable the simultaneous evaluation of multiple targeted therapies within a single cancer type, allowing several research questions to be answered within one design protocol. This approach is particularly useful in high-prevalence cancers characterised by molecular heterogeneity, as it facilitates biomarker-based patient stratification while maintaining focus on one disease entity. Nevertheless, umbrella trials often require collaboration across multiple institutions to support different treatment arms, increasing coordination complexity. Furthermore, inaccurate biomarker testing may increase the risk of false-positive or false-negative results, and many umbrella trials remain early-phase and exploratory in nature.
Why Do These Designs Matter for Pharma?
The U.S. Food and Drug Administration (FDA) has formally modernised guidance to the master protocol designs, including umbrella and basket trial designs, supporting their implementation in oncology drug development. This will result in earlier treatment signal detection, maximum use of the patient population, and will also give the chance for exploring treatment options for rare cancer cases.
Conclusion
Oncology trials are moving towards precision medicine, using basket and umbrella trial designs, which are considered innovative approaches that would be a turning point in oncology, and they will transform how therapies are developed and evaluated. Despite the challenges and statistical limitations of those models, researchers expect that those designs will play an increasing role in precision medicine.
| Basket Trial | Umbrella Trial | |
| Core Concept | One intervention tested across multiple cancer types sharing the same mutation. | Multiple interventions tested within one type of cancer based on different mutations. |
| Patient Grouping | Patients grouped by shared genetic mutation regardless of tumour origin. | Patients grouped by specific biomarkers within the same cancer type. |
| Advantages | Allows evaluation in rare cancers and rare mutations Cost-effective by combining evaluations into one trial. | Answers multiple research questions within one disease Enables simultaneous testing of targeted therapies. |
| Limitations | Small sample size and limited generalizability Limited statistical power due to small populations. | Requires complex coordination across multiple interventions Risk of false positives/negatives if biomarker testing is inaccurate. |










